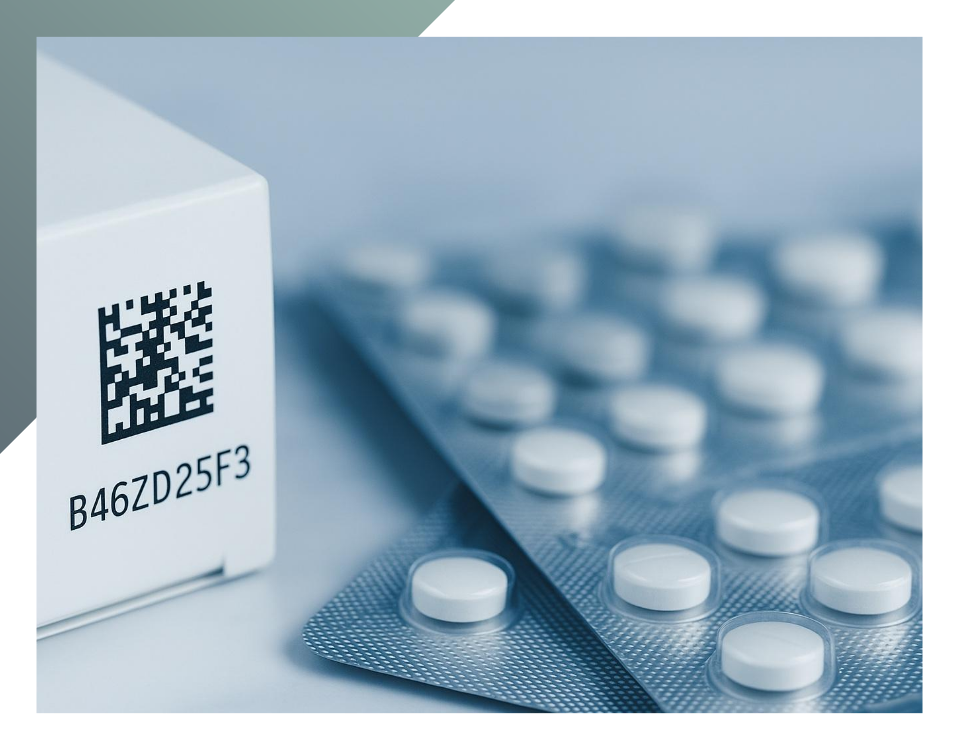
In the pharmaceutical packaging sector, serialization has by now become both a technical and regulatory standard. Beyond combating counterfeiting, it ensures the traceability of every single pack throughout the supply chain. However, implementing serialization effectively within well‑established industrial processes is far from straightforward.
Serialization: Regulatory Requirement and Strategic Lever
As set out by Directive 2011/62/EU and Delegated Regulation (EU) 2016/161, pharmaceutical companies are required to apply a GS1 Data Matrix to every saleable unit, containing unique identification data (GTIN, serial number, batch, and expiry date). In Italy, the derogation allowing the use of the traditional reimbursement sticker will expire by 2025, making the transition to the European system a priority also for local manufacturers. This transition, however, has a direct impact on automation, logistics, and data governance—key elements for all players involved in secondary packaging within a GMP environment.
Key Operational Challenges
What are the concrete impacts on the packaging industry?
1. Integration on existing lines. Retrofitting packaging lines with printing, verification, rejection, and aggregation modules requires mechatronic redesigns, testing, and validation activities. The main risk is introducing bottlenecks in critical production phases.
2. Code verification and quality control. OCR/OCV systems must ensure correct code readability, proper positioning, and real‑time traceability. Non‑compliant packages must be handled through automated rejection systems.
3. Cybersecurity and compliance. Since serialized data are a cornerstone of drug traceability, their management requires validated infrastructures compliant with 21 CFR Part 11 and GAMP5, fully integrated with company ERP/MES systems. In this context, data integrity in pharma becomes a crucial aspect.
Advanced Solutions and Reference Models
Some companies have invested in fully integrated serialization modules, combining high‑resolution printing, visual inspection, and multi‑level aggregation management. As a specialized player, Eurpack has developed dedicated serialization lines and Packaging Development Centers, where materials, formats, and compatibility with traceability systems are tested. Another practical example is the adoption of aggregated serialization, where individual units are associated with multipacks and pallets through unique coding structures. This approach improves logistical visibility and simplifies export operations—factors that are increasingly central within the pharmaceutical digital supply chain. Serialization is now an integral part of pharmaceutical packaging identity. Addressing it effectively requires scalable, interoperable, and regulatory‑compliant solutions. The key is to act early, turning a regulatory obligation into a competitive advantage.
How can we help you?
If you would like to learn more about our commitment to sustainability or have any questions about our projects and initiatives, we are here to provide you with all the information you need.